


Similar term(s): polychlorinated biphenyls.
Polychlorinated biphenyls (PCBs) are a family of 209 congeners of structurally similar organic chemicals, ranging from oily liquids to waxy solids. There are 12 PCBs that are dioxin-like and can similarly be toxic and non-toxic. One dioxin-like PCB is 3,4,4',5-Tetrachlorobiphenyl.
PCBs are synthetic and produced either as a singular congener, as a homogeneous group or as a mixture. They are non-flammable, stable, have a high boiling point and exhibit electrical insulating properties. As such, PCBs have been used as coolants and lubricants in transformers and other electrical equipment, as hydraulic fluids, and as plasticizers, pigments, dyes and carbonless copy paper ink. They are also generated and released into the environment as waste byproducts of chemical manufacturing and incineration.
Source: GreenFacts
Each PCB molecule contains two phenyl rings. A phenyl ring is a ring of 6 carbon atoms to which hydrogen atoms are attached. In PCBs, one or several chlorine atoms replace some of these hydrogen atoms. The two rings in a PCB molecule can rotate around the bond connecting them.
Depending on where the chlorine atoms are located, the two rings of a specific PCB will either:
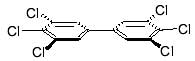
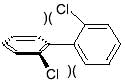
Coplanar PCBs are considered to be most toxic, based on combined health effects considerations. They are also referred to as “dioxin-like”.
Structure of dioxins:
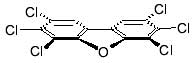
Source: GreenFacts
Congener - Dioxin(s) - Dioxin-like - Non toxic - Toxic
Deutsch: PCBs
Español: PCB
Français: PCB